Macrophages play a critical role in the initiation, maintenance, and resolution of inflammation. Function of these key immune cells can lead to both beneficial and detrimental outcomes in inflammation, therapeutic interventions that modulate macrophages may open new avenues for controlling inflammatory diseases.
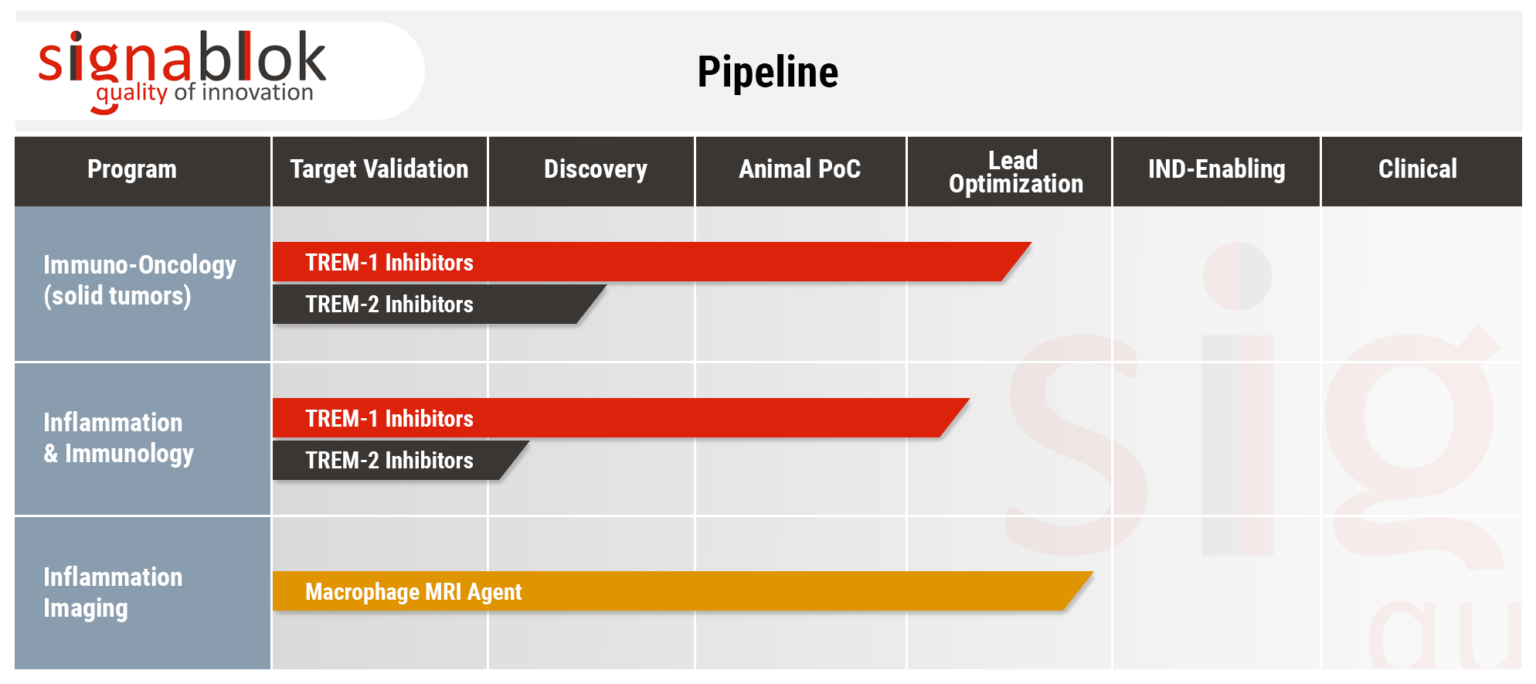
TREM-1, an inflammation-amplifying receptor, is overexpressed on macrophages upon inflammation and can serve as a checkpoint between a protective and detrimental immune response in virtually all inflammation-associated diseases including:
- cancer
- autoimmune diseases
- retinopathy
- atherosclerosis
- sepsis/acute respiratory distress syndrome (ARDS) and COVID
- other inflammatory diseases and conditions
Since the discovery of TREM-1 in 2000, the actual nature of the TREM-1 ligand(s) remains uncertain. In addition, different TREM-1 ligands are possibly involved in the pathogenesis of different inflammatory disorders. This emphasizes the hurdles that need to be overcome before TREM-1-targeting therapy can become a clinical reality.
We used the SCHOOL drug discovery platform to design the peptide sequence GF9 that employs a ligand-independent mechanism of TREM-1 inhibition. Due to the ability of GF9 to reach its site of action in the cell membrane from both inside and outside the cell, GF9 can be used either in a free form or formulated in SignaBlok's macrophage-specific lipopeptide complex for targeted intracellular delivery to macrophages.
Cell and animal studies have confirmed the high efficacy of SignaBlok's first-in-class ligand-independent TREM-1 inhibitory strategy in prevention and treatment of a variety of inflammation-associated diseases and disorders.
TREM-2 is expressed on newly differentiated and alternatively activated macrophages and attenuates macrophage activation. Recent studies demonstrated that TREM-2 plays a key role in regulation of antitumor immunity.
Further expanding on its technology platform, SignaBlok has launched a program focusing on a TREM-2-mediated modulation of immune response. The TREM2 program is currently in discovery phase.
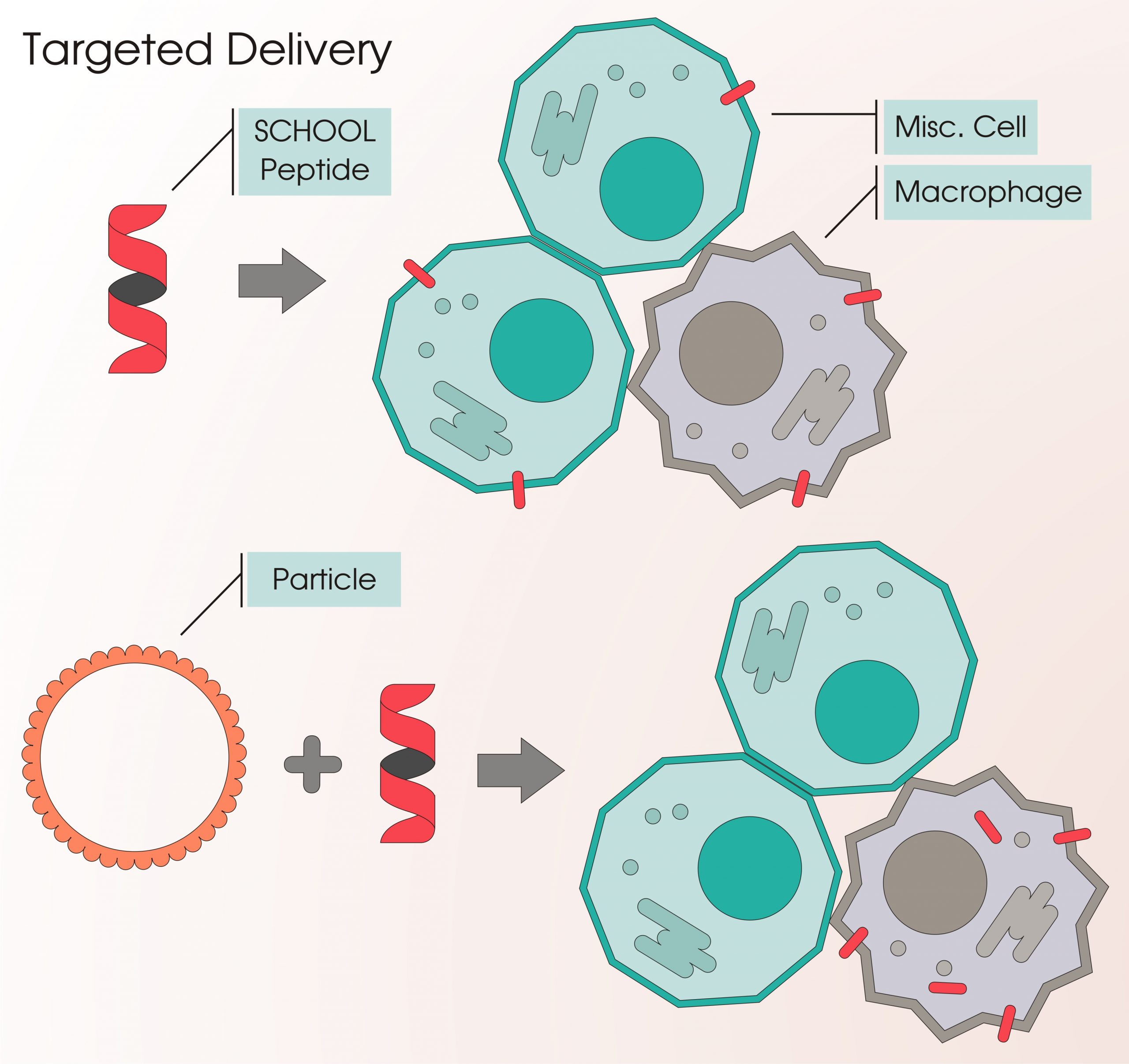
Targeted Delivery
Inspired by nature that uses lipoprotein complexes (lipoproteins) to transport lipids within the water-based bloodstream in a human body, SignaBlok’s founders created multifunctional lipopeptide complexes for targeted intracellular delivery of drugs and imaging agents to macrophages.
These particles were originally designed for targeted delivery of SignaBlok's TREM inhibitors and magnetic resonance imaging (MRI) contrast agents for macrophage modulation and imaging, but this versatile platform can be readily tuned to deliver a variety of imaging agents as well as therapies, ranging from small molecules to peptides and RNAs to sites of inflammation with applications in cancer, atherosclerosis, sepsis, rheumatoid arthritis, and other inflammation-associated diseases.