Imaging
Atherosclerosis, a leading cause of vascular disease, causes nearly 18 million deaths worldwide yearly with rupture of unstable (vulnerable) inflamed atherosclerotic plaques being a major reason. None of available non-invasive imaging modalities can detect and evaluate unstable plaques.
In addition, despite about 12 millions patients on lipid-lowering drugs only in the U.S. and about 300 clinical trials of novel lipid-lowering drug therapies in the U.S. annually, none of available non-invasive imaging modalities can monitor direct effect of treatment on atherosclerotic plaques.
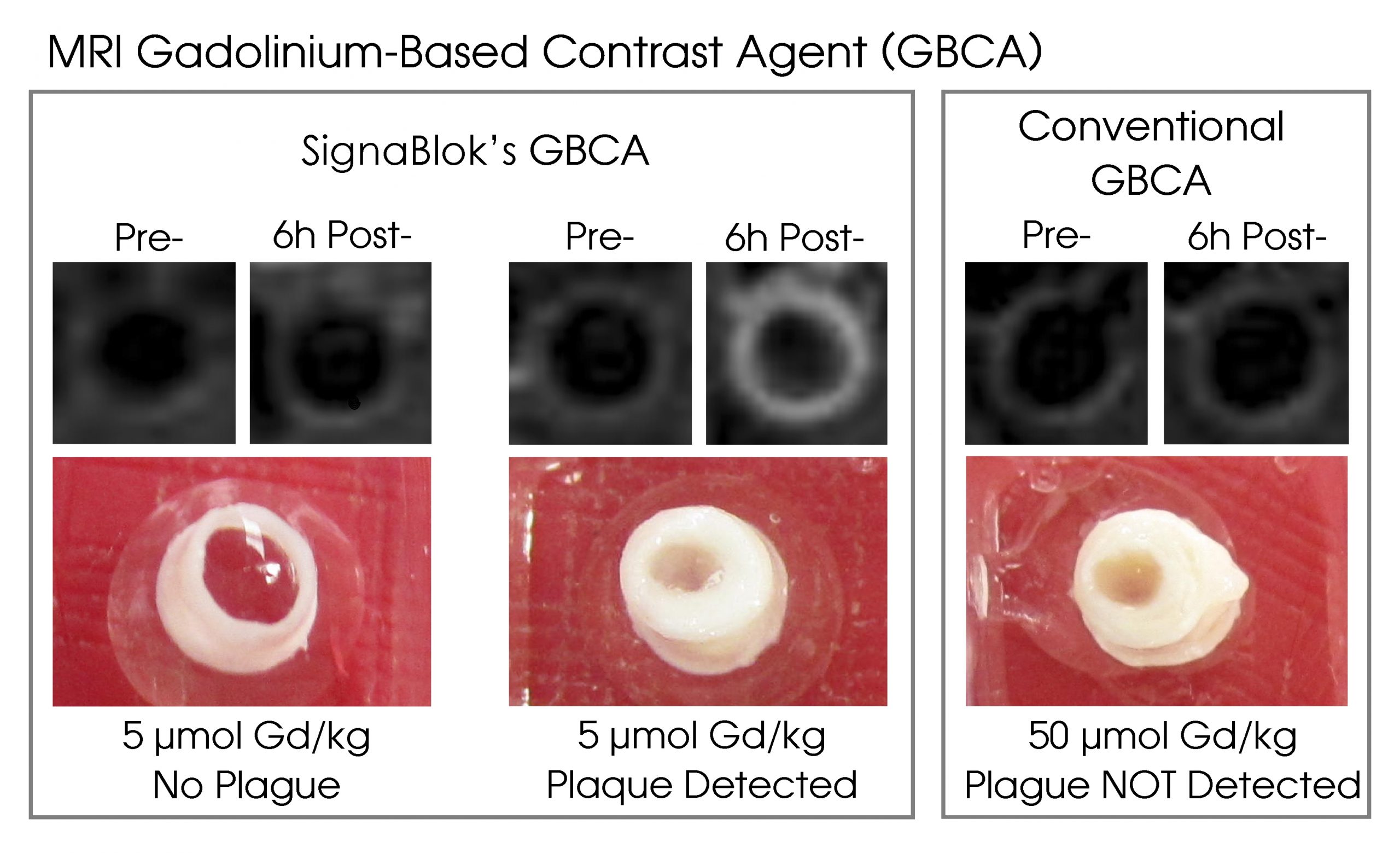
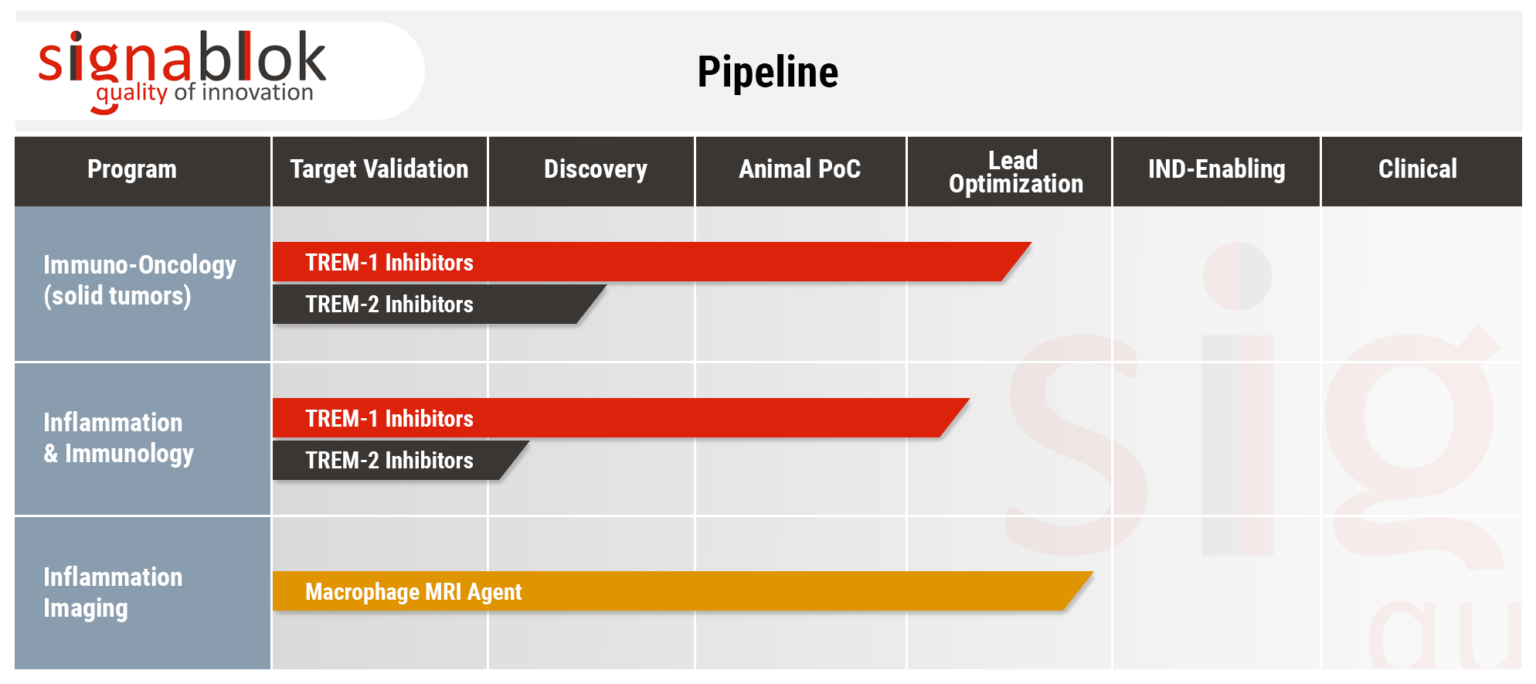
SignaBlok developed ultra high sensitivity (UHS) Gd-Based Contrast Agent (GBCA) for Magnetic Resonance Imaging (MRI) to detect inflammation in vivo with the main focus on atherosclerosis.
In addition to its unique ability to detect inflammation in vivo, this targeted UHS-GBCA is efficient at 20-fold lower Gd dose compared to those of current conventional GBCAs. Conventional GBCAs all have FDA's Black Box Warning but are still in use in over 30 million procedures of contrast MRI yearly worldwide due to lack of alternative. In addition to a significantly lower dose of Gd needed, hepatic clearance of SignaBlok's UHS-GBCA further diminishes known Gd renal toxicity.
Inflammation imaging in vivo using SignaBlok's UHS-GBCA is of diagnostic value in rheumatoid arthritis, cancer, liver diseases, Alzheimer's disease and other inflammatory diseases and conditions.
Treatment
TREM-1 has been recently identified as a major upstream proatherogenic receptor and as an upstream target in atherosclerosis, accordingly. TREM-1 levels are increased in atheromatous human atherosclerotic lesions and high circulating levels of soluble TREM-1 are significantly associated with major adverse cardiovascular events in patients with acute myocardial infarction, further suggesting clinical relevance of TREM-1 as a therapeutic target.
In preclinical models, pharmacological TREM-1 inhibition substantially reduces both atherosclerosis formation and progression.